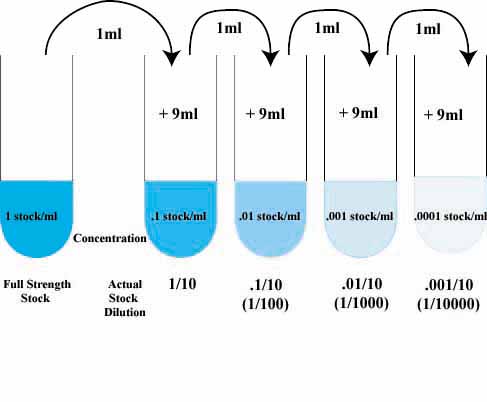
Serial Dilutions Practice
Dilution Problems
#1 - 10
#1 - 10
- Serial Dilution Table
- Serial Dilutions Practice Problems Answers
- 1.1.5 Serial Dilutions Practice Problems
- Serial Dilution Equation Practice
- Mi Serial Dilutions Practice Problems
- Serial Dilution Practice Lab
Dilutions are useful in science when making solutions or growing an acceptable number of bacterial colonies to count. There are three formulas used to work microbiology dilution problems: finding individual dilutions, finding serial dilutions, and finding the number of organisms in the original sample. Feb 17, 2016 A very quick lesson on how to do the math for serial and simple dilutions.
Problem #1: If you dilute 175 mL of a 1.6 M solution of LiCl to 1.0 L, determine the new concentration of the solution.
Solution:
M1V1 = M2V2(1.6 mol/L) (175 mL) = (x) (1000 mL)
x = 0.28 M
Note that 1000 mL was used rather than 1.0 L. Remember to keep the volume units consistent.
Problem #2: You need to make 10.0 L of 1.2 M KNO3. What molarity would the potassium nitrate solution need to be if you were to use only 2.5 L of it?
Solution:
M1V1 = M2V2(x) (2.5 L) = (1.2 mol/L) (10.0 L)
x = 4.8 M
Please note how I use the molarity unit, mol/L, in the calculation rather than the molarity symbol, M.
Problem #3: How many milliliters of 5.0 M copper(II) sulfate solution must be added to 160 mL of water to achieve a 0.30 M copper(II) sulfate solution?
Solution:
M1V1 = M2V2(5.00 mol/L) (x) = (0.3 mol/L) (160 + x)
5x = 48 + 0.3x
4.7x = 48
x = 10. mL (to two sig figs)
The solution to this problem assumes that the volumes are additive. That's the '160 + x' that is V2.
Problem #4: What volume of 4.50 M HCl can be made by mixing 5.65 M HCl with 250.0 mL of 3.55 M HCl?
Solution:
Here is the first way to solve this problem:
M1V1 + M2V2 = M3V3(3.55) (0.250) + (5.65) (x) = (4.50) (0.250 + x)
Where x is volume of 5.65 M HCl that is added
(0.250 + x) is total resultant volume
Serial Dilution Table
0.8875 + 5.65x = 1.125 + 4.50 x
1.15x = 0.2375
x= 0.2065 L
Total amount of 4.50 M HCl is then (0.250 + 0.2065) = 0.4565 L
Total amount = 456.5 mL
Here is the second way to solve this problem:
Since the amount of 5.65 M added is not asked for, there is no need to solve for it.M1V1 + M2V2 = M3V3
(3.55) (250) + (5.65) (x - 250) = (4.50) (x)
That way, x is the answer you want, the final volume of the solution, rather than x being the amount of 5.65 M solution that is added.
Problem #5: A 40.0 mL volume of 1.80 M Fe(NO3)3 is mixed with 21.5 mL of 0.808M Fe(NO3)3 solution. Calculate the molar concentration of the final solution.
Solution:
Let's use a slightly different way to write the subscripts:
M1V1 + M2V2 = M3V3
There is no standard way to write the subscripts in problems of this type.
Substituting:
(1.80) (40.0) + (0.808) (21.5) = (M3) (40.0 + 21.5)M3 = 1.45 M
Problem #6: To 2.00 L of 0.445 M HCl, you add 3.88 L of a second HCl solution of an unknown concentration. The resulting solution is 0.974 M. Assuming the volumes are additive, calculate the molarity of the second HCl solution.
Solution #1:
M1V1 + M2V2 = M3V3Serial Dilutions Practice Problems Answers
(0.445) (2.00) + (x) (3.88) = (0.974) (2.00 + 3.88)
0.890 + 3.88x = 5.72712
3.88x = 4.83712
MGT Sam Coupe. Mattel Intellivision. Memotech MTX512. 
x = 1.25 M (to three sig figs)
Solution #2:
1) Calculate moles HCl in 0.445 M solution:
(0.445 mol/L) (2.00 L) = 0.890 moles
2) Set up expression for moles of HCl in second solution:
(x) (3.88 L) = moles HCl in unknown solution
3) Calculate moles of HCl in final solution:
(0.974 mol/L) (5.88 L) = 5.73 moles
4) Moles of HCl in two mixed solutions = moles of HCl in final solution:
0.890 moles + [(x) (3.88 L)] = 5.73 molesx = 1.25 M (to three sig figs)
Problem #7: To what volume should you dilute 133 mL of an 7.90 M CuCl2 solution so that 51.5 mL of the diluted solution contains 4.49 g CuCl2?
Solution:
1) Find moles:
(4.49g CuCl2) (1 mole CuCl2 / 134.45 grams) = 0.033395 moles CuCl2
2) Find the molarity of the 51.5 mL of the diluted solution that contains 4.49g CuCl2:
(0.033395 moles CuCl2) / (0.0515 liters) = 0.648 M
3) Use the dilution formula:
M1.1.5 Serial Dilutions Practice Problems
1V1 = M2V2(7.90 M) (133 mL) = (0.648 M) (V2)
V2 = 1620 mL
You should dilute the 133 mL of an 7.90 M CuCl2 solution to 1620 mL.
Problem #8: If volumes are additive and 95.0 mL of 0.55 M KBr is mixed with 165.0 mL of a BaBr2 solution to give a new solution in which [Br¯] is 0.65 M, what is the concentration of the BaBr2 used to make the new solution?
Solution:
moles of Br¯ from KBr: (0.55 mol/L) (0.095 L) = 0.05225 mol
moles of Br¯ in final solution: (0.65 mol/L) (0.260 L) = 0.169 mol
moles Br¯ provided by the BaBr2 solution: 0.169 - 0.05225 = 0.11675 mol
BaBr2 provides two Br¯ per formula unit so (0.11675 divided by 2) moles of BaBr2 are required for 0.11675 moles of Br¯ in the solution.
molarity of BaBr2 solution: 0.058375 mol / 0.165 L = 0.35 M
Problem #9: 1.00 L of a solution is prepared by dissolving 125.6 g of NaF in it. If I took 180 mL of that solution and diluted it to 500 mL, determine the molarity of the resulting solution.
Solution:
1) Calculate moles of NaF:
125.6 g / 41.9 g/mol = 3.00 mol
2) Calculate moles in 180 mL of resulting solution:
3.00 mol in 1000 mL so 3 x (180/1000) = 0.54 mol in 180 mL
3) Calculate molarity of diluted solution:
0.54 mol / 0.50 L = 1.08 mol/L = 1.08 M
Problem #10: What is the molar concentration of chloride ions in a solution prepared by mixing 100.0 mL of 2.0 M KCl with 50.0 mL of a 1.50 M CaCl2 solution?
(Warning: there's a complication in the solution. It has to do with the CaCl2.)
Solution #1:
1) Get total moles of chloride:
KCl ⇒ (2.00 mol/L) (0.100 L) = 0.200 mol of chloride ionCaCl2 ⇒ (1.50 mol/L) (0.0500 L) (2 ions / 1 formula unit) = 0.150 mol of chloride ion.
The '2 ions / 1 formula unit' is the problem child. The solution is 1.50 M in calcium chloride, but 3.00 M in just chloride ion.
total moles = 0.200 mol + 0.150 mol = 0.350 mol Essential english vocabulary pdf.
2) Get chloride molarity:
0.350 mol / 0.150 L = 2.33 M
Solution #2:
Suppose you really wanted to use this equation:
Serial Dilution Equation Practice
M1V1 + M2V2 = M3V3
Set it up like this:
Mi Serial Dilutions Practice Problems
(2.00 mol/L) (0.100 L) + (3.00 mol/L) (0.0500 L) = (MSerial Dilution Practice Lab
3) (0.150 L)Note that the CaCl2 molarity is 3.00 because that is the molarity of the solution from the point-of-view of the chloride ion.